The stability of different intermediates during a reaction is important when studying the kinetics and final products of a reaction. The stability of different ions produced is reliant on the angle strain and steric effects as well as the possibility of methyl or a hydride shift.
Fridel Krafts
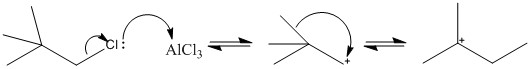
Fridel Krafts is a great example of when an ion can stabalise itself causing a change in product. This is due to the formation of the carbocation which can easily undergo a number of rearrangements. The examples below show how rearrangement can occur forcing the use of a more stable ion such as an acylation technique.
The following reaction mechanism is an example of how the rearrangement of the carbocation can occur.
Fridel Krafts is a great example of when an ion can stabalise itself causing a change in product. This is due to the formation of the carbocation which can easily undergo a number of rearrangements. The examples below show how rearrangement can occur forcing the use of a more stable ion such as an acylation technique.
The following reaction mechanism is an example of how the rearrangement of the carbocation can occur.
The HOMO orbital or the sigma bond between the methyl group and the carbon group. The empty p orbital (LUMO) interacts with the HOMO forming a tertiary carbocation.
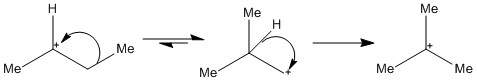
Secondary carbocations can form a tertiary cabocation through a methyl shift and then a hydride shift.
Secondary carbocations can form a tertiary cabocation through a methyl shift and then a hydride shift.
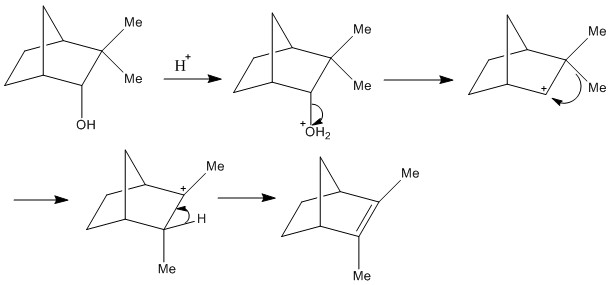
Wagner-Meerwein Rearrangements
Cation rearrangement is a kinetically accessible process. It can be a problem with some reactions but it can be useful when used synthetically. It is important to remember as with almost all of these that the reaction starts with the initial loss of the hydroxide group forming a cation. This catino can then undergo methyl shift.
Cation rearrangement is a kinetically accessible process. It can be a problem with some reactions but it can be useful when used synthetically. It is important to remember as with almost all of these that the reaction starts with the initial loss of the hydroxide group forming a cation. This catino can then undergo methyl shift.
This reaction is useful as there is little possibility of alternate reactions. The formation of a carbocation on a bridging group is highly unlikely as the carbocation is highly unstable as the cation cannot be planar. The formation of an alkene attached to the bridging group has the same problem and the alkene is very strained.
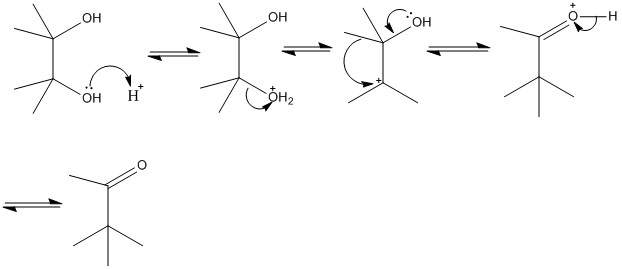
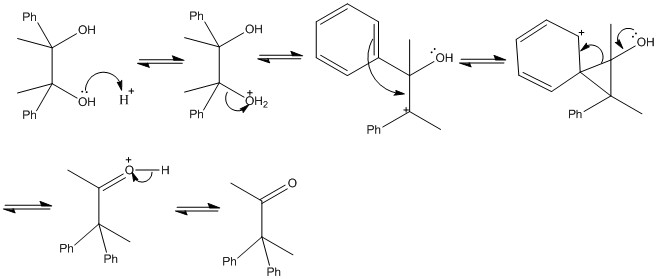
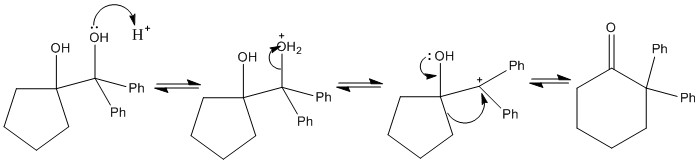
The pinacol rearrangement
Treatment of a 1,2-diol with acid can result in rearrangement. The reaction is names after pinacol, the trivial name for 2,3-dihydroxy-2,3-dimethylbutane.
The pinacol rearrangement
Treatment of a 1,2-diol with acid can result in rearrangement. The reaction is names after pinacol, the trivial name for 2,3-dihydroxy-2,3-dimethylbutane.
Choice of migrating group
As can be seen in the pinacol rearrangemtn one of the groups has to migrate. This is important when dervivng the final structure as there can be significant differences in the groups formed.
The phenyl group is much more likely to shift than the methyl group. This is due to an intermediate formed in the reaction. This is an important example of the intramolecular electrophilic substitution reaction.
As can be seen in the pinacol rearrangemtn one of the groups has to migrate. This is important when dervivng the final structure as there can be significant differences in the groups formed.
The phenyl group is much more likely to shift than the methyl group. This is due to an intermediate formed in the reaction. This is an important example of the intramolecular electrophilic substitution reaction.
Choice of carbocation
When the diol is not symmetric there can be a large difference in stabilities of the carbocation formed. This means that one of the carbocations will more likely form than the other and the reaction occur. The most stable carbocation should form this is important and it always the next stable carbo cation. Molecules cannot predict the future.
When the diol is not symmetric there can be a large difference in stabilities of the carbocation formed. This means that one of the carbocations will more likely form than the other and the reaction occur. The most stable carbocation should form this is important and it always the next stable carbo cation. Molecules cannot predict the future.
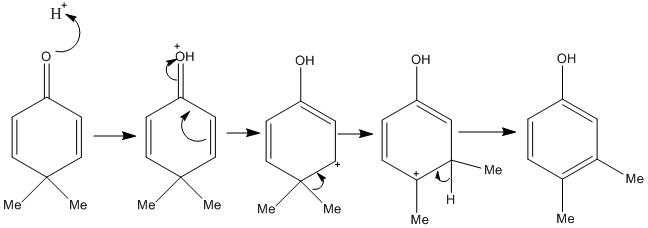
The dienone-Phenol Rearrangement
The dienone phenol rearrangement is an important one which can allow the movement of alkyl groups around a ring structure producing an aromatic compound at the end. The mechanism starts with protonation which causes a delocalisation of negative charge. This causes a methyl shift which then a less stable system to form. This can be made more stable by then losing a proton and forming a aromatic ring.
The dienone phenol rearrangement is an important one which can allow the movement of alkyl groups around a ring structure producing an aromatic compound at the end. The mechanism starts with protonation which causes a delocalisation of negative charge. This causes a methyl shift which then a less stable system to form. This can be made more stable by then losing a proton and forming a aromatic ring.
Where there are groups that are attached to the oxime carbon then the trans group is the migrating group. This is due to the HOMO LUMO pair being better pointed towards the trans bond then the cis bond.
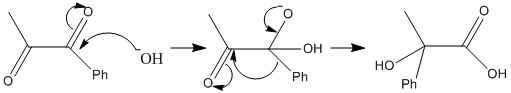
Benzylicc acid rearrangement
The rearrangement of a benzylic acid group is important and shows how the formation of a mesomeric carboxyl group is favoured.
Benzylicc acid rearrangement
The rearrangement of a benzylic acid group is important and shows how the formation of a mesomeric carboxyl group is favoured.
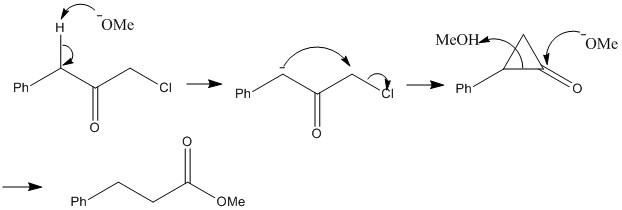
Favorskii rearrangement
It might be expected that the mechanism of the Favorskii reaction is similar to the cleavage of a C-X bond replaces involvement of the C=O bond. This involves the formation of a three centred ring which can then break again forming a different delocalised pattern causing a different product to form.
It might be expected that the mechanism of the Favorskii reaction is similar to the cleavage of a C-X bond replaces involvement of the C=O bond. This involves the formation of a three centred ring which can then break again forming a different delocalised pattern causing a different product to form.
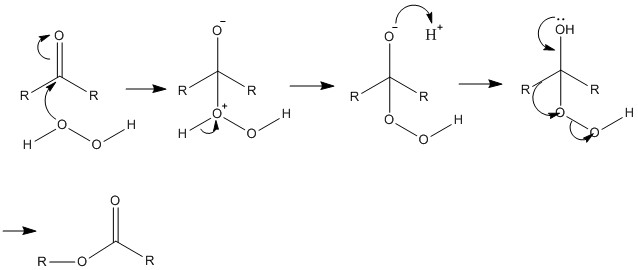
Baryer-Villiger Reaction
This reaction is a very important synthetic procedure as it can add an oxygen to a carbonyl group forming an ester. The insertion can be carried out using a peracid, with the most common reagent being m-CPBA.
This works by the oxygen bonding to the carbonyl group through the carbon. This causes a bond to form which can then be broken, this leads to migration of one of the alkyl groups go the positive charge.
This reaction is a very important synthetic procedure as it can add an oxygen to a carbonyl group forming an ester. The insertion can be carried out using a peracid, with the most common reagent being m-CPBA.
This works by the oxygen bonding to the carbonyl group through the carbon. This causes a bond to form which can then be broken, this leads to migration of one of the alkyl groups go the positive charge.